Introduction
SENS-401 is a first-in-class drug candidate to treat Sudden Sensorineural Hearing Loss (SSNHL). It is aiming at treating or protecting against inner ear lesions that lead to nerve degeneration and sensory hair cell loss. It is protected by a solid intellectual property with two patent families.



Structure
Diagram
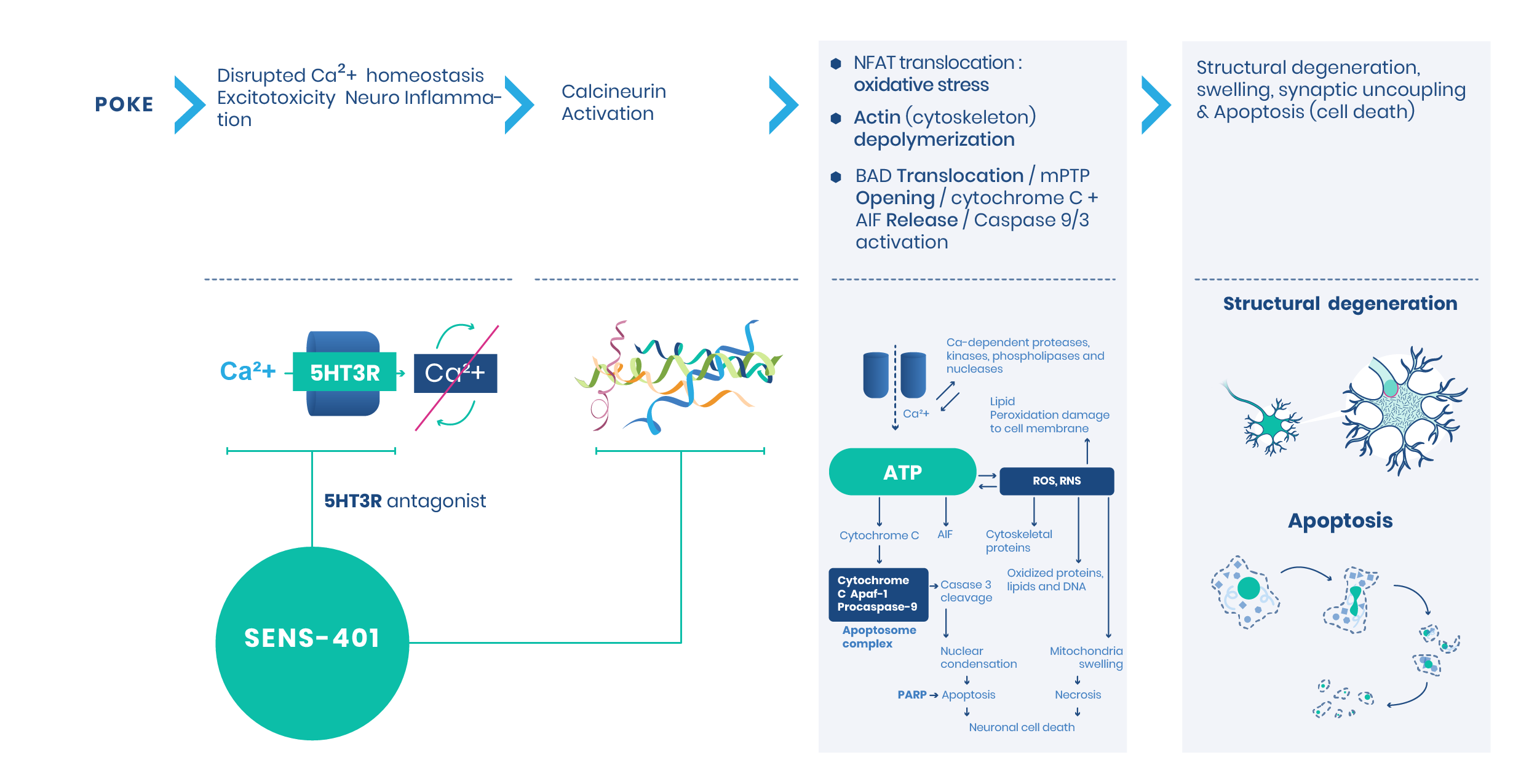
Promising trials

Introduction
SENS-401 is our lead candidate to treat sudden sensorineural hearing loss (SSNHL).
Key Opinion Webinar on SSNHL and SENS-401 as a potential treatment option
Listen to the presentation
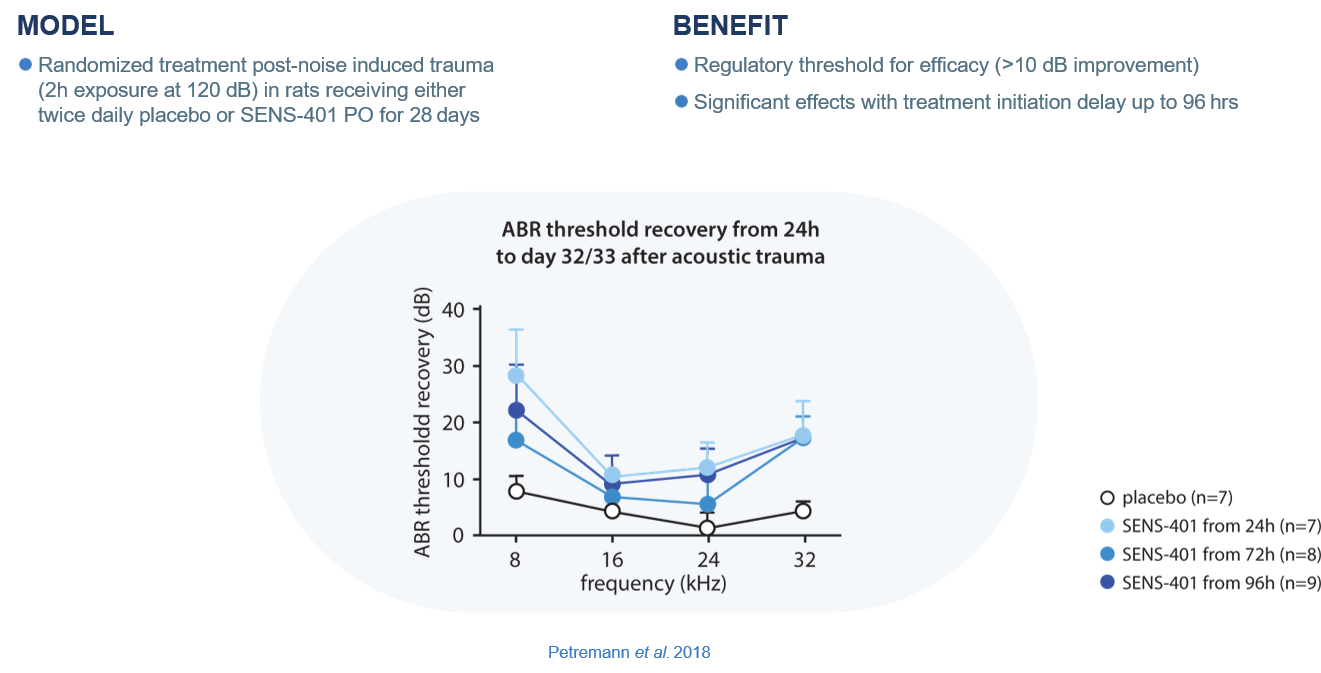
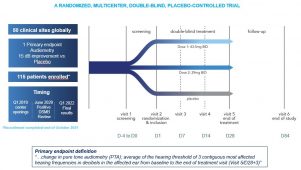
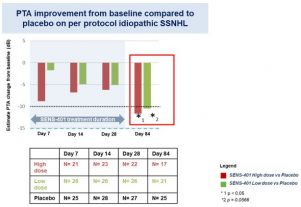
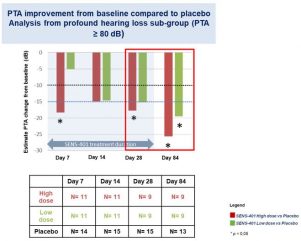
*
The PATRIOT consortium consists of Sensorion, the French Army Biomedical Research Center (IRBA), Institut Pasteur and Electronique du Mazet (Echodia), a French MedTech company focusing on hearing assessment and diagnostics. This project has a collaborative research and development structure in order to respond to the existing high unmet medical need in acute acoustic trauma. The PATRIOT consortium contributes to the development of SENS-401 as a treatment option, up to regulatory approval. The French government has awarded the PATRIOT consortium a ‘Structural Research and Development Project for Competitiveness’ (PSPC) grant with non-dilutive funding of up to €10.8m over the duration of the project. Sensorion could receive up to €5.6m. This grant will be used to finance the development of SENS-401 in SSNHL, including work by Institut Pasteur to identify response biomarkers. The commitment and active involvement of the French ministry of Armed Forces in this project emphasizes a clear unmet medical need and the necessity to have efficient diagnostics and therapeutic solutions. Auditory deficit is an invisible injury causing disabilities and potential social isolation.
Introduction
We are developing SENS-401 in the prevention of hearing loss due to Cisplatin-Induced Ototoxicity.
KOL webinar on CIO
Webinar final slides (English)
Listen to the presentation

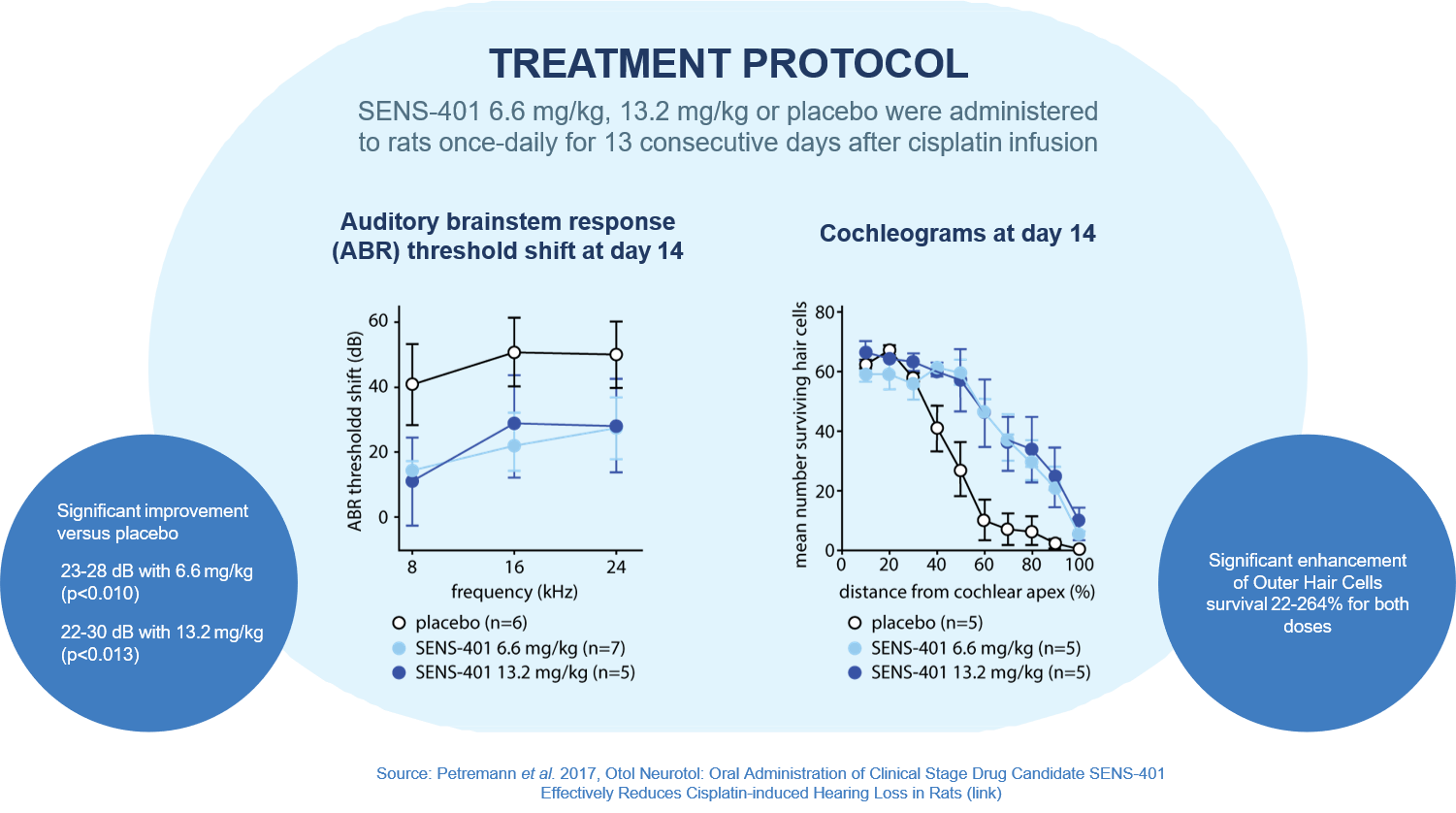
Introduction
We are investigating SENS-401’s otoprotective potential in patients with cochlear implants in collaboration with Cochlear, the global leader in implantable hearing solutions.



Results